AttachLifter and AttachGuider
by Rupp et al.
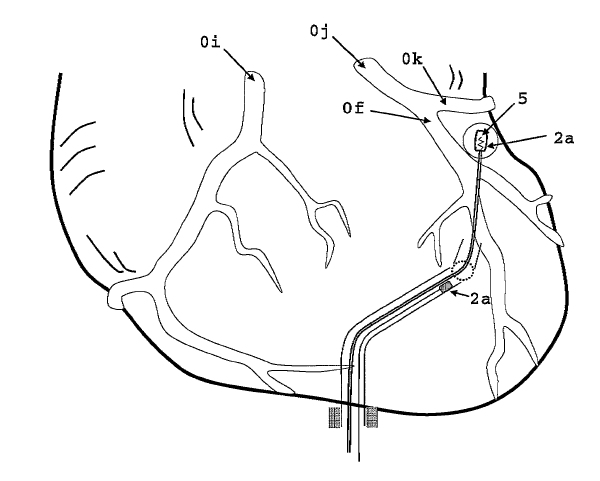
Access to the normal pericardial sac - no thoracoscopy, no lung deflation.
Epicardial ablation for rhythm management
Update 13/12/2011: EU patent on our key technology for minimal invasive pericardial access with the AttachLifter is granted.
Press release and in-licensing by TransMIT

How to develop devices for intrapericardial or epicardial access?
for a summary as PDF, see Rupp et al. AttachLifter
Our aims:
Multisite epicardial pacing for cardiac resynchronization therapy (CRT) after
i. minimal invasive access to the pericardial space (AttachLifter)
ii. intrapericardial navigation and mapping for best functional improvement (AttachGuider)
iii. epicardial lead implantation using controlled tissue attachment (principle of Attacher)
Contrary to current procedures for epicardial lead placement, the procedures do not require deflation of a lung and associated risks in frail patients. Lung deflation is required during thoracotomy (opening of the chest cavity with a large incision) and thoracoscopy (a small incision in the chest wall). Since the lung is partially deflated during thoracoscopy, the procedure cannot be done in patients whose lung function is poor. Lung deflation also puts a big load on the heart muscle.
In our minimal invasive approach, the surface of the heart can be reached via the subxiphoid procedure. In the context of the conventional pericardiocentesis, the subxiphoid approach is used routinely under local anaesthesia: "Dr. Maisch advocates the use of the subxiphoid approach to the puncture site, which requires fluoroscopy in a cardiac catheterisation suite and involves inserting a needle substernally via the left side of the scapula, at a 30 degree angle to the skin. During pericardiocentesis, the operator intermittently aspirates fluid and injects contrast medium. After aspiration, the needle is quickly replaced with a soft J-tip guidewire. After dilation the guidewire is replaced in turn by a pigtail catheter."
Instead of the needle which obviously cannot be used when pericardial effusion is absent (as in CRT patients), the PeriAttacher would be the preferred instrument, since it has a blunt end and cannot injure the heart surface.
| Key
terms: Attacher: our devices with the term "attach" are characterized by the controlled attachment of the tissue to an instrument during a particular procedure, e.g. electrode implantation on the surface of the heart (epicardium). When tissue attachment is lost for whatever reason, this is recognized and the procedure is stopped until the surgeon regains tissue attachment. AttachLifter: a follow-up of the Attacher which obviously fails in the case of thickened (fat deposition or fibrosis) pericardium. The device uses a novel procedure for entering the pericardial space. AttachGuider: a device for guiding instruments, e.g. during navigation inside the pericardial space. The AttachGuider differs from conventional endoscopes and is made for navigation in a cavity without a wall for deflecting the endoscope head. AttachImplanter: a device which permits secure implantation of a pacing lead in the epicardium. It is a follow-up of the simple srew-in lead described below and prevents loosening of the lead. The electrode is also relieved from the mechanical stress imposed by the wall movement. Cannot be disclosed yet. |
Bundle branch block and resynchronization - a challenge
The pump function of the heart requires the coordinated excitation of the left and right ventricle. Excitation, i.e. the heart beat, starts in the sinus node of the right atrium and the impulse moves from the atria to the ventricles through the AV node and then into the right and left bundle branches which lead to the apex of the heart. From the viewpoint of physiology, the heart should contract first at the apex, thereby pressing the blood from the apex to the base where it is ejected into the aorta and pulmonary artery. A "bundle branch block" is a slowing or interrution of the impulses and can occur in the left (LBBB) and right ventricle (RBBB). Any disordered excitation of the heart chambers is associated with depressed pump performance. Cardiac resynchronization therapy (CRT) should ideally restore the synchrony of the pumping action of the left and right ventricle, thereby improving the overall function of the heart. Bundle branch block is often caused by another disease such as coronary artery disease, inflammatory heart disease and particularly during progression of heart failure.
The disordered excitation in bundle branch block can be visualized by the excellent ECGsim program: "If a bundle of the Purkinje system is blocked, the depolarization of the region of the heart that is served by this bundle is delayed. To simulate a left bundle branch block, select a node and range. Set the depolarization time of the selected node to about 180 ms. You will see all typical effect of a left bundle branch block such as elongation of the QRS-complex" In the right Figure, a LBBB is present which results in delayed excitation and the characteristic ECG. The yellow vertical line in the ECG (bottom Figure) marks the time of the heart cycle. The ECG in blue represents a normal heart and the ECG in red represents a heart with LBBB.

"Heart failure: the ironic failure of success"
This statement by the late Professor R.E. Beamish (Can J Cardiol 1994;10:603) summarizes the current therapy for heart failure. Drugs prolong survival but can too often not prevent final failure. Cardiovascular diseases have been identified as "public health enemy No. 1" by the World Health Organization. The search for reliable preventive methods should therefore vigorously be pursued. In particular, novel therapeutic targets are required beyond pharmaceuticals. In this context, problems associated with a disturbed propagation of the heart rhythm in the two ventricles have been addressed by medical device companies. The beneficial effects of CRT have prompted inclusion of this therapeutic option into current guidelines for congestive heart failure management. Nonetheless, there is increasing evidence that the beneficial action of resynchronization could be optimized by carefully selecting the location where stimulating of the left or right ventricle occurs. The aim should be to mimick physiological excitation.
Right ventricular pacemaker - how can a life saving strategy be improved?
Despite intense research, adequate treatment of cardiac rhythm disorders resulting in an impaired heart function remains a major medical need. While drug treatment of ventricular tachyarrhythmias has remained disappointing, the implantation of right ventricular pacemakers has evolved as life saving strategy. Nonetheless, the question arises whether right ventricular pacing could be optimized by not only stimulating the right but also the left ventricle, again mimicking the normal physiologal excitation of the heart. This issue is addressed in the MINERVA: MINimizE Right Ventricular Pacing to Prevent Atrial Fibrillation and Heart Failure conducted by Medtronic Bakken Research Center: "Several authors have shown that, in patients with intact AV conduction, unnecessary chronic RV pacing can cause detrimental effects such as AF, left ventricular (LV) dysfunction and congestive heart failure. These findings arose the hypothesis that the non-physiologic nature of ventricular pacing may result in electrophysiological and LV remodeling changes that have potentially deleterious long-term effects." In this study, right ventricular pacing is used only when required: "The MVP mode, present in the Medtronic pacemaker EnRhythm, provides atrial based pacing with ventricular backup. It operates in true AAI(R) mode, it provides ventricular backup in case of a single conduction loss and converts to DDD(R) mode in case of persistent loss of AV conduction. Aim of this study is to test the impact of the MVP pacing mode and atrial preventive and antitachycardia pacing therapies on the reduction of a composite clinical outcome composed by any death, permanent AF, cardiovascular hospitalizations."
The question behind is whether right ventricular pacing may have similar effects to LBBB and is associated with dyssynchrony of the left and right ventricular pumping action. This important issue is addressed also in the pioneering BioPace study by Dr. R.C. Funck et al. Patients assigned to biventricular pacing received the Model 5510 Frontier 3 x 2 (St Jude Medical Inc) triple-chamber pacemaker or newer models. Depending on whether sinus rhythm or chronic atrial fibrillation is present, patients randomized to the RV pacing group received conventional single or dual-chamber pulse generators manufactured by St Jude Medical Inc.
"Although it is unclear whether the negative effects of RV pacing LBBB are comparable, and whether they depend on the presence and the degree of structural heart disease, one may hypothesize that RV pacing may have similar effects to LBBB."
"Despite the deleterious effects of cardiac dyssynchrony and the positive effects of cardiac resynchronization therapy, patients with high-degree atrioventricular block continue to receive desynchronizing right ventricular (RV) pacing systems. Although it is unclear whether the negative effects of RV pacing and left bundle branch block (LBBB) are comparable, and whether they depend on the presence and the degree of structural heart disease, one may hypothesize that RV pacing may have similar effects to LBBB. In the BioPace trial, the long-term effects of RV pacing vs. biventricular pacing will be prospectively compared in 1200 pacemaker patients with high likelihood of mostly paced ventricular events,
regardless of whether in sinus rhythm or in atrial fibrillation (AF). After echocardiographic examination of left ventricular (LV) function, patients will be randomly assigned to the implantation of an RV vs. a biventricular pacing system and followed for up to 5 years. Primary study endpoints are survival, quality of life (QoL), and the distance covered in a 6-min hall walk (6-MHW) at 24 months after implantation. Secondary endpoints are QoL and the 6-MHW result at 12 months after implantation, hospitalizationrate, LV dimensions, LV ejection fraction, and the development of chronic AF and other adverse events." Cited from Europace (2006) 8, 629–635.
"Biventricular Pacing for Atrioventricular Block to Prevent Cardiac Desynchronization" is in our opinion a pioneering step towards "physiological" excitation of the heart. In case of an advantage of biventricular pacing, the next pioneering step is to assess whether the used coronary sinus approach with few options for lead placement can be improved by individualized epicardial lead placement.
In view of the high incidence of heart failure and unresolved issues with right ventricular pacing, efforts should be made to better achieve the goal of CRT, i.e. an optimized synchronization of the pumping action of heart chambers as outlined also by R.K. Shepard and K.A. Ellenbogen in 2007:
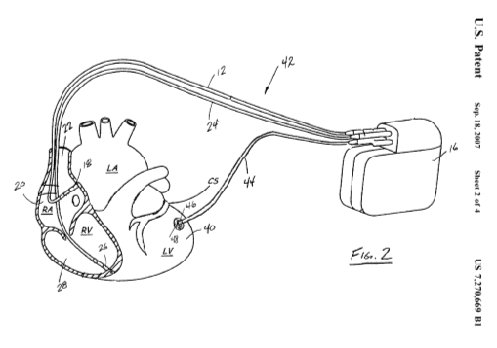
While the currently preferred procedure for resynchronization involves insertion of a pacing lead into a left ventricular tributary vein of the coronary sinus, there is a clear need for minimally invasive techniques leading to optimized resynchronization. The need for novel interventions for CRT has also been pointed out also by Jasbir S. Sra of Medtronic Inc in United States Patent 7270669 issued on September 18, 2007: Epicardial lead placement for bi-ventricular pacing using thoracoscopic approach:
"Despite considerable progress in the management of CHF, it remains a major health problem worldwide. It is estimated that there are 6-7 million people with CHF in the United States and Europe alone and approximately 1 million new patients are diagnosed with CHF every year.
Despite significant advances in the treatment of CHF using various pharmacological therapies, the quality-of-life in patients with CHF remains poor as these patients are frequently hospitalized, and heart failure in these patients is a common cause of death. In addition, there are significant long-term care costs associated with this problem. Many patients with advanced CHF have a conduction system disease that may play a role in worsening cardiac function. One frequently noted conduction abnormality is left bundle branch block (LBBB), which is present in about 29% of patients with CHF. The presence of LBBB delays left ventricular ejection due to delayed left ventricular activation.
Pacing therapies have been introduced in an attempt to improve cardiac function in patients diagnosed with CHF. Cardiac resynchronization, in which bi-ventricular pacing is performed, has shown beneficial results in patients with CHF and LBBB. During bi-ventricular pacing, in addition to the standard right atrial and right ventricular leads, an additional lead is positioned in the coronary sinus. This additional lead is advanced into one of the branches of the coronary sinus overlaying the epicardial surface of the left ventricle. Since the lead is advanced through the coronary sinus, the potential placement positions for the lead are severely limited" (see Fig.1).

Although bi-ventricular pacing has shown beneficial results, numerous problems are associated with this technique.
One such problem is the amount of time required for a physician to insert the lead into the desired location on the left ventricle. Further, the placement of the lead on the left ventricle is limited to sites that provide adequate pacing and sensing signals. Further, cannulation of the coronary sinus is often difficult due to either the rotation of the heart or the presence of an enlarged right atrium or Thebesian valve. Further, the placement of a lead on the surface of the left ventricle though the coronary sinus cannot be carried out in some patients with prior bypass surgery or with coronary sinus stenosis. Finally, the coronary sinus lead provides an oftentimes unstable placement and can become detached after installation.
Therefore, a need exists for an alternative approach that allows for the easier placement of the left ventricle lead and an apparatus for positioning such a lead in the desired location"; see Fig. 2 (above text in quotation marks and figure 1 and figure 2 are from United States Patent 7270669).
In this invention, the socalled "stingray technique" is used for inserting a left ventricular lead in a thoracoscopic approach that places the lead at a specific, known location on the epicardial surface of the left ventricle. Although very promising from a physiological viewpoint, the procedure has the disadvantage of not being minimally invasive requiring lung deflation. However, patients who need CRT often have also an impaired lung function and procedures should thus be preferred which do not have the inherent risk of further deterioration of lung performance. Since such procedures are to our knowledge not available, we initiated a broad research project focussing on minimally invasive access to the pericardial space (subxiphoid approach) followed by epicardial lead implantation.
Another limitation of the transvenous approach relates to the number of leads which can be implanted in coronary veins which for obvious reasons is very limited, in particular with respect to the anatomical placement. The study of C. Leclercq et al. is important since it provides evidence that the conventional approach can indeed by improved by LV multisite pacing.
OBJECTIVES: We compared the effects of triple-site versus dual-site biventricular stimulation in candidates for cardiac resynchronization therapy. BACKGROUND:
Conventional biventricular stimulation with a single right ventricular (RV) and a single left ventricular (LV) lead is associated with persistence of cardiac
dyssynchrony in up to 30% of patients. METHODS: This multicenter, single-blind, crossover study enrolled 40 patients (mean age 70 +/- 9 years) with
moderate-to-severe heart failure despite optimal drug treatment, a mean LV ejection fraction of 26 +/- 11%, and permanent atrial fibrillation requiring
cardiac pacing for slow ventricular rate. A cardiac resynchronization therapy device connected to 1 RV and 2 LV leads, inserted in 2 separate coronary sinus
tributaries, was successfully implanted in 34 patients. After 3 months of biventricular stimulation, the patients were randomly assigned to stimulation for
3 months with either 1 RV and 2 LV leads (3-V) or to conventional stimulation with 1 RV and 1 LV lead (2-V), then crossed over for 3 months to the alternate
configuration. The primary study end point was quality of ventricular resynchronization (Z ratio). Secondary end points included reverse LV remodeling,
quality of life, distance covered during 6-min hall walk, and procedure-related morbidity and mortality. Data from the 6- and 9-month visits were combined to
compare end points associated with 2-V versus 3-V. RESULTS: Data eligible for protocol-defined analyses were available in 26 patients. No significant
difference in Z ratio, quality of life, and 6-min hall walk was observed between 2-V and 3-V. However, a significantly higher LV ejection fraction (27 +/- 11% vs.
35 +/- 11%; p = 0.001) and smaller LV end-systolic volume (157 +/- 69 cm(3) vs. 134 +/- 75 cm(3); p = 0.02) and diameter (57 +/- 12 mm vs. 54 +/- 10 mm; p =
0.02) were observed with 3-V than with 2-V. There was a single minor procedure-related complication. CONCLUSIONS: Cardiac resynchronization therapy
with 1 RV and 2 LV leads was safe and associated with significantly more LV reverse remodeling than conventional biventricular stimulation.
Optimized placement of pacing leads requires thus the intrapericardial approach
Where should the epicardial pacing lead be placed? As pointed out by Mair et al: For optimal biventricular pacing, the LV lead has been found to be best placed in the area where optimal concordance is achieved between the LV pacing site and the site of the most delayed LV wall. For anatomical or technical reasons, the placement of the LV lead via the coronary sinus at the intended target area of the LV is often not possible.
An option for avoiding these drawbacks is the surgical implantation of the left ventricular lead under direct vision. In the study of Mair et al., three epicardial lead implantation techniques were described that are less invasive. In patients with advanced heart failure and left bundle branch block, epicardial left ventricular leads for biventricular pacing were implanted with: (1) left lateral mini-thoracotomy; (2) a video-assisted thoracoscopy approach using lead implantation tools; and (3) a robotically enhanced telemanipulation system. It was concluded: Epicardial lead implantation for biventricular pacing is feasible with all 3 surgical techniques. Each method allows optimal lead implantation under direct vision and therefore reduces the incidence of nonresponders resulting from suboptimal lead placement. Of importance is in this respect also the development of a testing tool for assessing if and how cardiac "re-synchronization" by pacing may be achieved.
The challenge: epicardial pacing in a minimal invasive approach without lung deflation
Cardiac resynchronization therapy has evolved as a valuable non-pharmacological treatment option for patients with advanced congestive heart failure, impaired left ventricular systolic function and intraventricular conduction abnormalities. We believe that the benefit for these patients could greatly be improved by multisite epicardial pacing of the ventricles involving a minimally invasive access to the pericardial space which does not require the adverse deflation of the lung. In view of the increasing burden of heart failure and the relative lack of success in preventing heart failure, we initiated an extensive research program for accessing the normal pericardial space (in the absence of pericardial effusion) for navigating within the pericardial space and for multisite epicardial pacing.
The necessity of a technique for epicardial lead implantation which is less invasive than the current procedures involving thoracotomy, thoracoscopy and port-based placement can be deduced also from the recent ACC/AHA/HRS 2008 Guidelines for Device-Based Therapy of Cardiac Rhythm Abnormalities: "The choice of surgical procedure appears to influence hospital morbidity. Surgical approaches for placement of LV epicardial leads include left thoracotomy, left thoracoscopy, and robotically assisted port-based placement. Thoracotomy in fragile patients with heart failure has been associated with bleeding, stroke, hypotension, and arrhythmias (470,476). In contrast, thoracoscopic and robotic approaches have been reported to be associated with minimal morbidity and may be preferred (472,473,475). These less invasive procedures generally require 60 to 90 minutes of operative time and a mean hospital stay of 4 to 5 days (472). However, not all patients are candidates for minimally invasive or robotic procedures. Subjects who have undergone prior thoracotomy or sternotomy operations may have limited pericardial/epicardial accessibility."
Since our procedure does not require lung deflation, it overcomes a major problem in the above socalled "minimal invasive" procedures, i.e. lung deflation in fragile patients.
|
Our
aims:
Multisite epicardial pacing for cardiac resynchronization therapy (CRT) after i. minimal invasive access to the pericardial space (PeriAttacher) ii. intrapericardial navigation and mapping for best functional improvement (AttachGuider) iii. epicardial lead implantation using controlled tissue attachment (principle of Attacher)
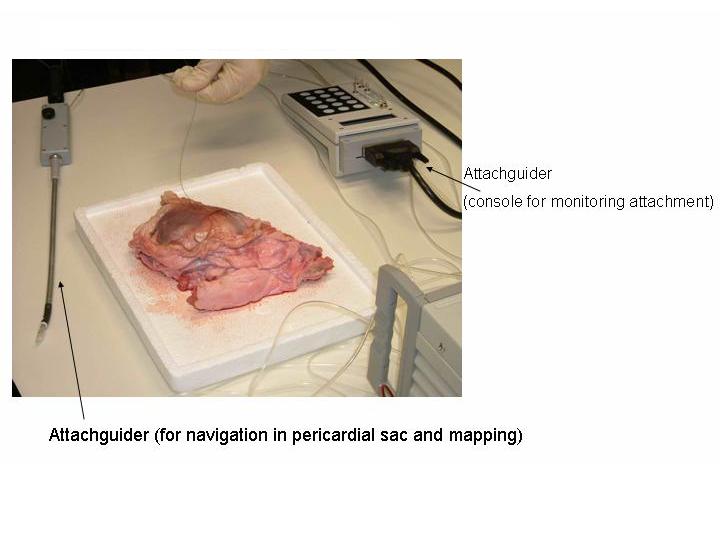
We have currently 3 patent applications for devices which provide access to the normal pericardial sac and intrapericardial navigation. Currently only our first two devices, the PeriAttacher and the AttachGuider can be disclosed. The devices use the principle of the Attacher, i.e. the attachment of tissue is monitored before and during any tissue manipulation (e.g. puncturing, implantation of screw-in lead etc). Tissue attachment is monitored by an increase in negative pressure which can be displayed by various means (therefore also the term "Attacher"). A pressure recording showing the increase in vacuum or negative pressure upon attachment is shown below:  The setup required is simple and requires a regular suction pump, a control console for monitoring tissue attachment and the PeriAttacher (please note, the device shown is an early prototype not suitable for thickened fatty pericardium).  The principal steps of accessing the pericardial sac involve (in the picture below from left to right): Attachment of the pericardium to the PeriAttacher and puncture of the pericardium with a needle (left), introduction of a guide wire located in the needle used in the puncturing step (middle) and dilation of the opening using dilators with successively increasing diameter (right). When the opening is large enough, other instruments can be introduced into the pericardial space, e.g. the AttachGuider for epicardial lead implantation or in case of pericardial effusion, biopsies could be taken and drugs instilled. 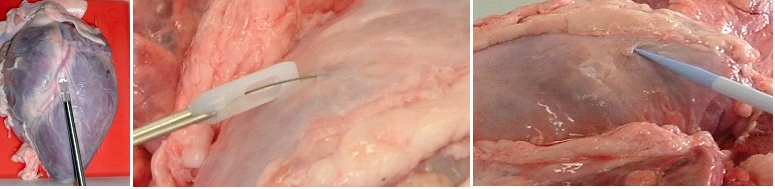 Socio-economic relevance of novel interventions targeting rhythm disorders in heart failure. Heart failure is a major cause of morbidity and mortality leading to steadily increasing health care expenditure. Our current research focuses on the treatment of a dysregulated gene expression of cardiomyocytes (4) and prevention of an adverse remodeling of the extracellular matrix arising from activation of the renin-angiotensin-aldosterone system. While these disorders can be treated with specific drugs, cardiac function is depressed further in about 30% of patients with heart failure by abnormalities in the electrical conducting system. In patients with dilated cardiomyopathy, up to 14% of the patients are candidates for resychronization therapy (5). An intraventricular conduction delay or bundle branch block (LBBB) occurs particularly in dilated hearts which causes the two ventricles to beat in an asynchronous manner. This asynchrony reduces the pump efficiency of the ventricles. Cardiac resynchronization therapy coordinates the pump function by pacing both ventricles simultaneously. Classic VVI (ventricular) pacemakers pace only the right ventricle. Data from a meta-analysis of studies on biventricular stimulation shows a significant reduction in all-cause mortality (6). In view of the fact that heart failure causes more hospitalizations than all forms of cancer combined, an improved minimal invasive implantation of epicardial pacing leads is expected to have a marked socio-economic impact. Current resynchronization procedures? A two-faced Janus Head? Epicardial pacing with optimized lead placement - but not minimal invasive! Minimally invasive pacing in a coronary vein - but not optimized lead placement! The proper pumping action of the
heart depends on a coordinated excitation of the
heart chambers. In the case of electrical
conduction abnormalities, biventricular
stimulation should ideally provide a procedure for
correcting any asynchrony and thus impaired
function of the two ventricles. Since an
impaired pump function is a major determinant of
ventricular tachycardias and sudden death (2),
preservation of pump performance remains an
important target.
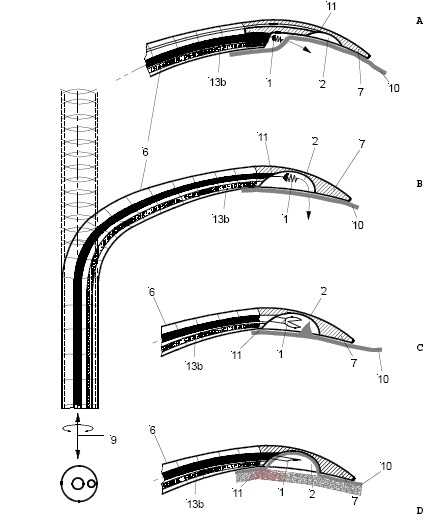
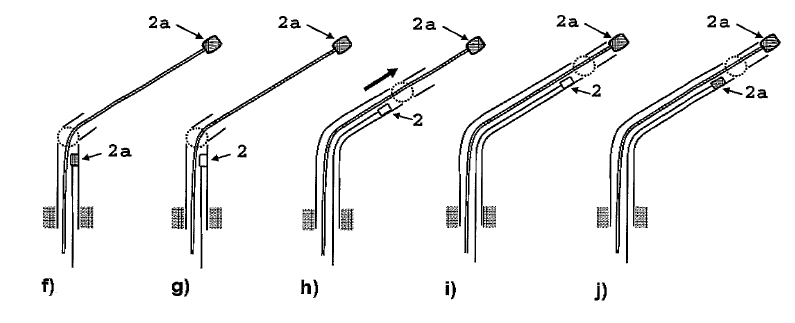
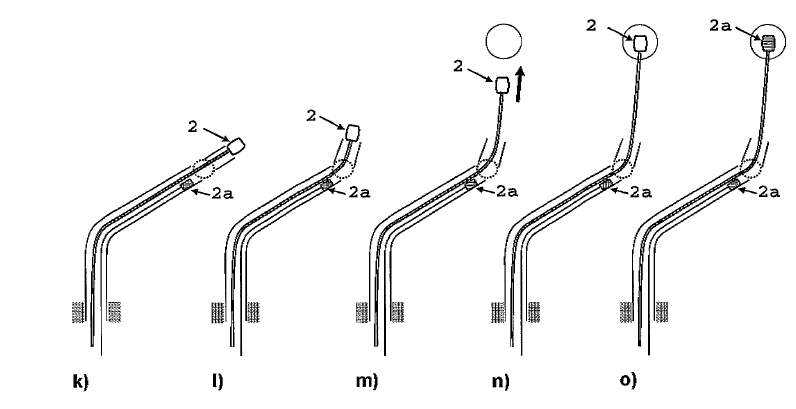
The biventricular pacing differs from the conventional pacemaker which stimulates only the right side of the heart and relies on the normal conduction system for transferring the pulse to the left ventricle. Biventricular pacing ideally requires a pacing lead on a specific location of the left ventricle which can a priori not be predicted. The best placement depends on various anatomical and physiological factors such as fibrosis and ischemia which can also not be predicted. Mapping for best functional improvement is, therefore, required. This can best be achieved by epicardial lead implantation which until now requires, however, a surgical intervention which by itself raises the risk for the patient. The surgical lead implantation involves thoracotomy with rib spreading and lead placement on the epicardium which results in good sensing and pacing thresholds. Unfortunately, the procedure is associated with morbidity particularly in frail patients. Although robotic technology has allowed a refinement of the thoracotomy, it requires extensive investments. Alternatives were, therefore, developed which were less invasive but suffered from the fact that mapping for best functional improvement cannot be done. Due to the noninvasivenesss of the technique, the standard approach became the percutaneous implantation involving insertion of the lead in a left lateral coronary vein through the coronary sinus ostium under fluoroscopic control. Technical difficulties can, however, arise from not reaching the desired coronary vein, failure of proper fixation negatively affecting the pacing or sensing thresholds. A coronary sinus-lead implantation can also fail and require conversion to epicardial leads involving left-lateral thoracotomy. It has been shown by Reichart et al. (3) that epicardial leads revealed better long-term results than transvenous coronary sinus lead implantation (lower stimulation-threshold after 12 months, no failure). In fact, when taking into account the number of patients with unsuccessful implantations and patients whose leads become dysfunctional during follow-up, the clinical failure may approach 50% (7). The current status of coronary sinus lead implantation with active fixation involving leads from Medtronic, Boston Scientific/Guidant, BioSpace, Biotronik, St. Jude Medical is given by Nägele et al. (Europace, 2007): " Aims Coronary sinus (CS) lead implantation is a technically challenging procedure owing to variable vein anatomies and a high dislocation rate. Therefore, CS lead technology has undergone evolutionary changes during the last 10 years. The mode of fixation has been a passive one up to now. We want to describe our first clinical experience with the newly available active fixation lead 4195 in terms of dislocation rate and stability of thresholds compared with conventional models. Methods and results From 1999 to February 2007, we implanted 403 CS leads in 368 patients. Leads were categorized into three different groups on the basis of their fixation mechanism: straight (Easytrak I and Situs OTW; n = 54), curved (Attain 4193 and 4194, Corox, Aescula, Situs ULD; n = 308), and active (Attain 4195; n = 41). Operative and follow-up data were prospectively noted and checked for significance between groups during the first 3 months after implantation. Kaplan–Meier analysis of long-term lead function was also performed. Straight and curved CS leads suffered from significantly more dislocations compared with active fixation (P < 0.001). The active fixation lead (4195) has a stable threshold over time compared with a significant rise after 24 h and thereafter in straight (62%) and curved leads (20%). However, retraction of an active fixation CS lead may be a difficult issue as outlined in two cases requiring pullback of a 4195 lead owing to phrenic nerve stimulation (one unsuccessful despite vigorous traction). Conclusion The active fixation lead 4195 using retention lobes yielded stable thresholds over time and seems to be superior to conventional leads in terms of dislocation. However, extraction may be a difficult or even impossible task."Therefore, the need for improving epicardial lead implantation procedures keeps growing. Devices for epicardial lead placement have been developed by Medtronic and Enpath. Steroid eluting epicardial leads have also been developed, by e.g. St. Jude Medical. A major limiting factor of the epicardial approach remains the required invasive thoracotomy in patients with already depressed heart performance and often impaired lung function. Minimally invasive epicardial lead implantation - our aims Minimal invasive approaches for implanting epicardial leads after mapping for best functional improvement represent, therefore, an urgent medical need. In our research, we followed a two-step approach, i.e. first the development of devices for minimally accessing the pericardial space (PeriAttacher and follow-up devices) and secondly devices for intrapericardial navigation and mapping for best functional improvement (AttachGuider using the principle of the Attacher, i.e. tissue manipulation only during controlled attachment). In view of the many unresolved technical problems, we addressed at first the question whether a minimal invasive access to the pericardium can indeed be achieved. For accessing the pericardial sac with effusion, a system referred to as "PerDUCER" has already been developed by Comedicus Inc. The device involves manual suction with a syringe for attaching the pericardium to the head piece prior to puncture with a needle. Although this procedure has been validated in pigs, it was less successful in patients. In a study performed by Prof. B. Maisch at the Department of Cardiology, Philipps University of Marburg a successful puncture of the pericardium with access to the pericardial sac was achieved in 2 out of 6 patients (8). This failure rate might not be unexpected taking into account that the attachment to the pericardium was achieved only by manual suction with a syringe and that in parallel to the suction, the needle had to advanced for puncturing the attached pericardium. Any loss of attachment is expected to result in failure of pericardial puncture. Also thickened pericardium would fill out the suction head and the needle would thus not enter the pericardial space.  How to develop devices for a minimal invasive access to the pericardial space? A prototype of the Attacher for puncturing the pericardial sac (named PeriAttacher for this purpose) is shown below. A fiberscope was attached which could be used for inspecting the tissue before puncturing the pericardium (picture taken during demonstration of the “11284A Vaskular-Fiberskop” of Karl-Storz Endoscopy, Inc., Tuttlingen, Germany). Please note the successful guide wire insertion. Guide wires were kindly provided by EPflex .  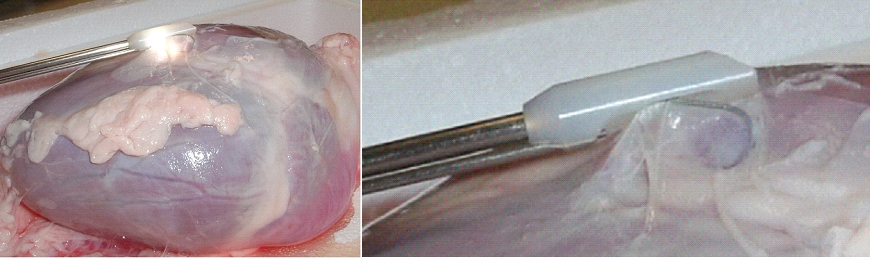  Device for a minimal invasive access to the pericardial space with thickened pericardium  Our aims: Multisite epicardial pacing for cardiac resynchronization therapy (CRT) after i. minimal invasive access to the pericardial space (PeriAttacher) ii. intrapericardial navigation and mapping for best functional improvement (AttachGuider) iii. epicardial lead implantation using controlled tissue attachment (principle of Attacher) Only for the purpose of demonstrating the implantation of an epicardial lead using our "controlled attachment" approach (as specified in the Attacher patent application), the procedure is shown using a very early prototype which is derived from a flexible endoscope. 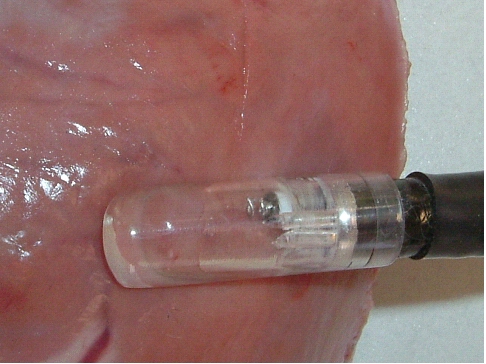 Suction head for "controlled attachment
".
Head of the instrument has been attached to the
surface of the heart (epicardium) and the screw-in
electrode advanced. Note, the pericardium has been
removed in this heart.
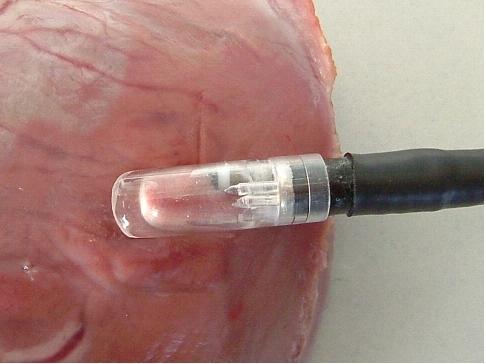 Head of the instrument is still attached
and
the electrode has been screwed into the myocardium.
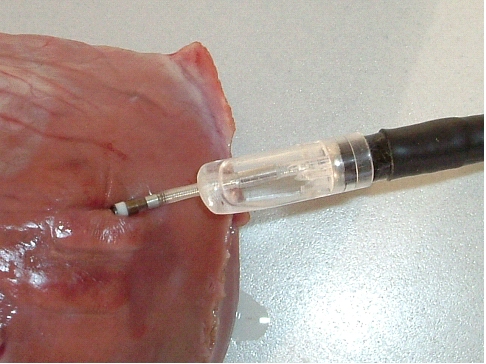 Head of the instrument has been detached
while the electrode (this particular lead is just
used for demonstrating the screw-in mechanism, it is
not to be meant as a working pacing lead) remains
securely screwed into the myocardium. The lead is
implanted approx. tangentially to the epicardial
surface. In the usual approach, the lead is
implanted perpendicually to the surface.
During myocardial contraction, the forces tend
to loosen such a lead which is implanted in parallel
with the force of contraction. When the lead is
implanted perpendicularly to these forces, the risk
of loosening of the lead and associated tissue
inflammation/fibrous tissue formation is expected to
be much less pronounced.
Are epicardial leads useful for long-term pacing? A convincing answer to this question has been given in 2006 by B. Ector et al. Epicardial pacing: a single-centre study on 321 leads in 138 patients "OBJECTIVE: This study presents the long-term outcome of 321 epicardial leads in 138 patients. METHODS AND RESULTS: All leads were Medtronic CapsureEpi model 4965 steroid eluting leads. The 1-, 3-, and 5-year patient survival was 91%, 83% and 77%, respectively.Twenty-seven patients died. In 25/27 deaths a pacing-related death could be excluded. Strangulation of the heart by an abandoned epicardial lead was the cause of death in one child. One other patient died suddenly at the age of 3 years. Failures occurred in 57 of 321 epicardial leads (18%). For all 321 leads, the 1-, 3- and 5-year freedom from failure was 91%, 85% and 71%, respectively.The cumulative proportion of patients without any lead defect was 85% after I year, 76% after 3 years and 62% after 5 years. The percentage of patients without serious adverse events at 1, 3, and 5 years was 97%, 91%, 85%, respectively. Lead fracture was the cause of failure in 15 leads of 9 patients. An important increase in pacing threshold occurred in 35 leads of 30 patients. Other failures were: diaphragmatic stimulation, infection, excessive traction and strangulation. Eighteen failures were repaired by 11 surgical interventions in 9 patients.Thirty-nine defects were corrected non-invasively in 31 patients. CONCLUSIONS: The use of steroid-eluting epicardial leads has proven to be anadequate option. In paediatric cardiology, the epicardial approach remains an indispensable tool for achieving a life-long pacing." Therefore, epicardial leads should be useful also for CRT. Our
aims:
Multisite epicardial pacing for cardiac resynchronization therapy (CRT) after i. minimal invasive access to the pericardial space (PeriAttacher) ii. intrapericardial navigation and mapping for best functional improvement (AttachGuider) iii. epicardial lead implantation using controlled tissue attachment (principle of Attacher)
How to develop devices for intrapericardial navigation? for a summary as PDF, see Rupp et al. AttachGuider After accessing the pericardial sac, a flexible fiberscope could be used for reaching predefined parts of the surface (epicardium) of the heart chambers (picture taken during demonstration of the “11282D1 Flexibles Vaskular-Fiberskop” of Karl-Storz Endoscopy, Inc., Tuttlingen, Germany). In this heart, the pericardium is thin but can be thickened by fat deposition or excessive collagen formation. 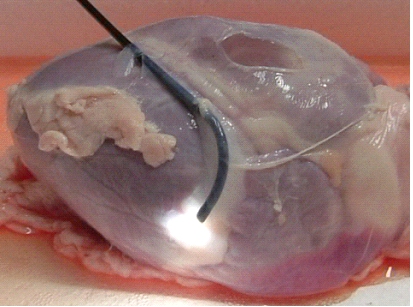   This principle was used for designing the AttachGuider which represents an endoscope which is different from the ones in clinical use. For precise intrapericardial navigation, the device has to maintain a position from which further movement can be initiated. The fixed location can be achieved by various means, in particular by two suction heads (with controlled attachment; outer device preferably to the pericardium, inner device to the epicardium which permits also pacing) or by extension of a balloon in the intrapericardial space.  Please note that the AttachGuider indeed differs in various aspects from the related art, in particular from the patent specifications EP 1 547 514 A1; US 6,007,482; WO 2006/123397 where a steerable endoscope is equipped with an inflatable balloon at the endoscope head for fixation of the endoscope. One might argue that the AttachGuider resembles the devices of patent specification EP 1 547 514 A1, where a flexible "insertion assisting tool" is pushed to the limit state onto the insertion section of the flexible endoscope. In this specification, the insertion assisting tool is directed by the movement of the steerable endoscope. This is completely different from the AttachGuider where the outer device directs the inner flexible device (or instrument). Furthermore, both the outer and the inner device of the AttachGuider carry means of attachment which can be of the type of a balloon or a suction head. While the balloon type of fixation is common, a suction head has also been described (WO 99/53827). However, all these devices require a canal, i.e. the intestine canal, which directs the endoscope head. Therefore, the AttachGuider differs from prior art which will be made clear in the upcoming US application of the AttachGuider PCT application. Since the AttachGuider can be equipped with a suction head for pacing at a given location, mapping for best functional improvement can be achieved during intrapericardial navigation. After identifying the best location, a permanent pacing lead can be implanted as shown in principle above. This procedure can be repeated and thus multiple leads could be implanted, i.e. multisite left ventricular pacing which would not be possible with the standard coronary sinus approach. Nonetheless, the procedures involved do not require lung deflation. Also because the extent of epicardial fat can vary, a directed intrapericardial movement is an essential part of the envisaged procedure. As reviewed by S.W. Rabkin (Obesity Reviews 2007;8:253–261), the greatest amount of epicardial fat is seen over the lateral right ventricular wall followed by the anterior wall with little present posteriorly. Since the electrical resistance or impedance of fat is much higher than that of muscle, leads implanted solely in fat tissue require a higher voltage stimulation threshold. Preferably, the tip of the lead enters the myocardium while the remaining part of the lead is securely fixed (screw-in lead) in the fatty tissue overlaying the muscle tissue. By this way, less damage of viable muscle tissue occurs. During movement to a location for mapping for functional improvement or epicardial lead implantation, the separate movements of the outer and inner device of the AttachGuider are monitored and displayed on the control console (in this case an LCD). The standalone mobile system is based on a microcontroller board (using FORTH).  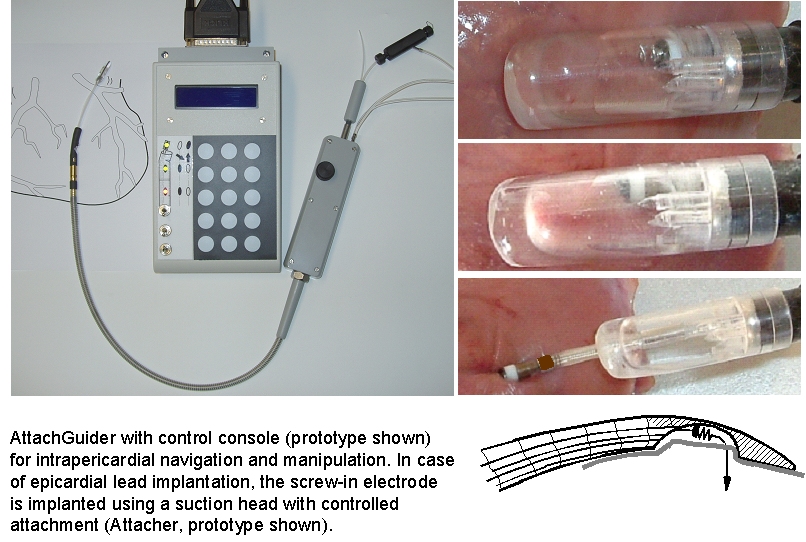 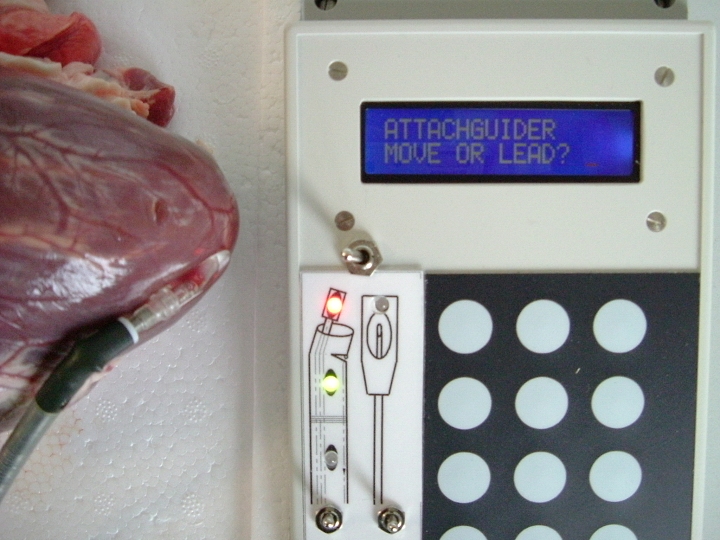 The prototype control console (based
on the TDS2020F
embedded FORTH
computer from Triangle
Digital Services which has been used e.g.
for data
logging) permits also opening and closure
of electrically driven miniature vacuum valves and
displays the status of the suction heads. Procedural instructions for the
user are displayed on an LCD. Alternatives
of the out-of-production TDS2020F could be the PDQ board
using the keypad/display
wildcard or the legacy product QED
Product Design Kit from Mosaic
Industries, the HC11 CPU based
POBox
from New Micros, the ARM
LPC2132 microcontroller board from ByVac
or the MPE
LPC2468 PowerBoard
from
MicroProcessor Engineering. Forth
was used since it provides an easy solution to the
turnkey application, is well structured and
flexible: Forth tutorials
and by Leo Brodie the classic Starting
Forth and the very enjoyable Thinking
Forth - A Language and Philosophy for Solving
Problems
   The preferred version for fixation during intrapericardial navigation involves controlled attachment to the pericardium from the inside of the pericardial sac. This is done using our standard procedure for controlled attachment to tissue. Two prototypes of a suction head are shown below. Either a firm material can be used (top) or a flexible material (as used in balloon catheters) can be used (bottom). The performance of these suction heads has been greatly improved by us and a patent application has recently been submitted.  An early prototype of a flexible version of the AttachGuider is also shown which permits directed movement in the pericardial space. In this version, the fixation during directed movement is achieved by a controlled extension of a balloon instead by a suction head. Attachment or fixation by balloon inflation is again electronically monitored and displayed.  Intrapericardial echocardiography and various ablation procedures The upcoming devices permit intrapericardial navigation which would be very useful also for e.g. intrapericardial echocardiography and various ablation procedures.Horowitz et al. assessed the potential of intrapericardial echocardiography (Heart Rhythm 2006;3:1275-82): "Percutaneous intrapericardial echocardiography during catheter ablation: a feasibility study.Horowitz BN, Vaseghi M, Mahajan A, Cesario DA, Buch E, Valderrábano M, Boyle NG, Ellenbogen KA, Shivkumar K. UCLA Cardiac Arrhythmia Center, Division of Cardiology, Department of Medicine, David Geffen School of Medicine at UCLA, Los Angeles, California 90095, USA. BACKGROUND: Percutaneous pericardial access, epicardial mapping, and ablation have been used successfully for catheter ablation procedures. OBJECTIVES: The purpose of this study was to evaluate the safety and feasibility of closed-chest direct epicardial ultrasound imaging for aiding cardiac catheter ablation procedures. METHODS: An intracardiac ultrasound catheter was used for closed-chest epicardial imaging of the heart in 10 patients undergoing percutaneous epicardial access for catheter ablation. All patients underwent concomitant intracardiac echocardiography and preprocedural transesophageal echocardiography. Using a double-wire technique, two sheaths were placed in the pericardium, and a phased-array ultrasound catheter was manipulated within the pericardial sinuses for imaging. RESULTS: Multiple images from varying angles were obtained for catheter navigation. Notably, image stability was excellent, and structures such as the left atrial appendage were seen in great detail. No complications resulting from use of the ultrasound catheter in the pericardium occurred, and no restriction of movement due to the presence of the additional catheter in the pericardial space was observed. Wall motion was correlated to voltage maps in five patients and showed that areas of scars correlated with wall-motion abnormalities. Normal wall-motion score correlated to sensed signals of 4.2 +/- 0.3 mV (normal myocardium >1.5 mV), and scores >1 correlated to areas with signals <0.5 mV in that territory). CONCLUSION: Intrapericardial imaging using an ultrasound catheter is feasible and safe and has the potential to provide additional valuable information for complex ablation procedures." An ultrasound catheter (ACUSON AcuNav™ 8F ultrasound catheter from Siemens Medical Solutions) could be used also during the access procedure to the pericardial sac. Robotic implantation of CRT and defibrillator rhythm management systems It is only few years back since cardiac surgeons in Frankfurt successfully completed robotic implantation of left ventricular electrodes for bi-ventricular pacing, after failure to access the coronary sinus approach. An update on robotic implantation of resynchronization and defibrillator rhythm management systems is given by G. Frank & O. Tyers (2006). Epicardial lead implantation following our minimally invasive approach with the PeriAttacher and AttachGuider appears as an alternative to robotic lead implantation and to robotic devices such as the HeartLander
which is a miniature
mobile robot to facilitate minimally invasive
beating-heart intrapericardial therapies which has
been developed by The
Robotics Institute, Carnegie Mellon
University, Pittsburgh.
Why is the conventional needle approach not adequate? Even in the presence of large pericardial effusion, the PeriAttacher should be advantageous when compared with the conventional needle. Pericardial effusion can accurately be assessed by cardiac magnetic resonance (CMR) (for avi file with better resolution, see Dr. Peter Alter, Heart Center Marburg) who pioneered in developing CMR-based wall stress calculation, the calculation of "isostress" curves for risk assessment and the assessment of cardiac contractility based on CMR derived presure-volume loops of the heart. A heart with pericardial effusion (right) is compared with dilative cardiomyopathy (center) and a normal heart (left). It is often assumed that a large pericardial effusion precludes the risk of puncturing the epicardium in the conventional needle approach. The CMR clearly demonstrates that this is not the case, even in a large pericardial effusion, the epicardium touches the pericardium on the front of the chest, i.e. the location where the needle is forwarded. Clearly, it depends on the skills of the cardiologist to avoid puncturing the epicardium and preventing life-threatening cardiac tamponade. In this condition, the use of the PeriAttacher and its follow-up device (not yet disclosed) would represent a safe alternative which excludes any risk of puncturing the epicardium. 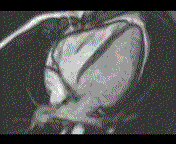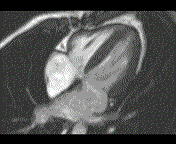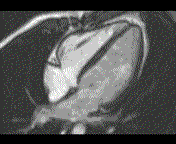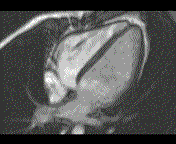 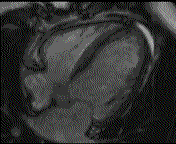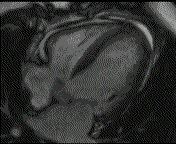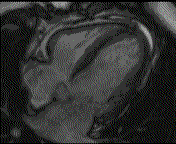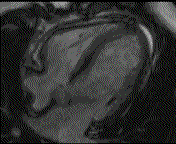 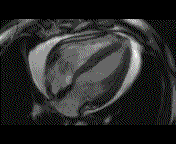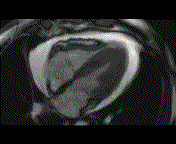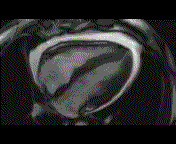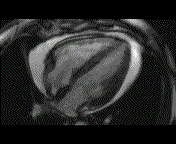 © P. Alter © P. AlterAre there also other routes to the pericardial space? Our devices have also to be seen in the context of the recent approach by S.R. Mickelsen et al. "Transvenous Access to the Pericardial Space: A Novel Approach to Epicardial Lead Implantation for Cardiac Resynchronization Therapy" involving a transvenous access to the pericardial space which, however, is associated with acute pericardial effusion during the implantation procedure: "Background:
Percutaneous
access
to the pericardial space (PS) may be useful for
a number of therapeutic modalities including
implantation of epicardial pacing leads. We have
developed a catheter-based transvenous method to
access the PS for implanting chronic medical
devices.Methods: In eight
pigs, a transseptal Mullins sheath and
Brockenbrough needle were introduced into the
right atrium (RA) from the jugular vein under
fluoroscopic guidance. The PS was entered
through a controlled puncture of the terminal
anterior superior vena cava (SVC) (n = 7) or
right atrial appendage (n = 1). A guidewire was
advanced through the transseptal sheath, which
was then removed leaving the wire in PS. The
guidewire was used to direct both passive and
active fixation pacing leads into the PS. Pacing
was attempted and lead position was confirmed by
cine fluoroscopy. Animals were sacrificed
acutely and at 2 and 6 weeks. Results:
All
animals survived the procedure. Pericardial
effusion (PE) during the procedure was
hemodynamically significant in four of the eight
animals. At necropsy, lead exit sites appeared to
heal without complication at 2 and 6 weeks. Volume
of pericardial fluid was 10.8 ± 6.2 mL and
appeared normal in four of the six chronic
animals. Moderate fibrinous deposition was
observed in two animals, which had exhibited
significant over-procedural PE. Conclusions:
Access
to the PS via a transvenous approach is
feasible. Pacing leads can be negotiated into
this region. The puncture site heals with the
lead in place. Further development should focus
on eliminating PE and performing this technique
in appropriate heart failure models."
References (1) Maisch B, Seferovic PM, Ristic AD,
Erbel R, Rienmuller R, Adler Y, Tomkowski WZ,
Thiene G, Yacoub MH. Guidelines on the diagnosis
and management of pericardial diseases executive
summary; The Task force on the diagnosis and
management of pericardial diseases of the European
society of cardiology. Eur Heart J 2004;
25:587-610.
(2) Grimm W, Christ M, Bach J, Muller HH, Maisch B. Noninvasive arrhythmia risk stratification in idiopathic dilated cardiomyopathy: results of the Marburg Cardiomyopathy Study. Circulation 2003; 108:2883-2891. (3) Mair H, Schütz A, Warncke K, Schmöckel M, Nollert G, Brömsen J, Reichart B. Epicardial leads are superior to coronary sinus leads in biventricular pacing. The Thoracic and Cardiovascular Surgeon 2003; V220 (abstract). (4) Turcani M, Rupp H. Etomoxir improves left ventricular performance of pressure- overloaded rat heart. Circulation 1997; 96:3681-3686. (5) Grimm W, Sharkova J, Funck R, Maisch B. How many patients with dilated cardiomyopathy may potentially benefit from cardiac resynchronization therapy? Pacing Clin Electrophysiol 2003; 26(1 Pt 2):155-157. (6) Salukhe TV, Dimopoulos K, Francis D. Cardiac resynchronisation may reduce all-cause mortality: meta-analysis of preliminary COMPANION data with CONTAK-CD, InSync ICD, MIRACLE and MUSTIC. Int J Cardiol 2004; 93:101-103. (7) Steinberg JS, Derose JJ. The rationale for nontransvenous leads and cardiac resynchronization devices. Pacing Clin Electrophysiol 2003; 26:2211-2212. (8) Maisch B, Ristic AD, Rupp H, Spodick DH. Pericardial access using the PerDUCER and flexible percutaneous pericardioscopy. Am J Cardiol 2001; 88:1323-1326. |
The Attacher and follow-up devices
have been developed at the Department
of Internal Medicine and Cardiology of the
Heart Center and the Technical Development Plant
of the Medical
Center and Medical Faculty of the Philipps
University
of
Marburg
The team involved in the production of the AttachLifter and related tools: Prof. Dr. Heinz Rupp Prof. Dr. Bernhard Maisch Dr. Thomas P. Rupp, M.D. Michael Koch Ekkehard Schüler Hermann Schön Karin Rupp Patent application and marketing is in cooperation with TransMIT (for the Transfer Center of the Philipps University) 
 Patents are required for our devices because they are the prerequisite for the development by major companies. Considerable investment is required for the production of devices certified for use in patients. We are greatly indebted to Dr. Peter Stumpf of TransMIT. Without his excellent writing of the patent applications, our devices would remain in the laboratory stage. For information on the AttachLifter and related tools, please contact Prof. Dr. Heinz Rupp For information on live demonstration of the AttachLifter and follow-up tools, please contact Dr. Thomas P. Rupp For information on the pending patent of the Attacher and follow-up devices, particularly the AttachGuider, please contact directly Dr. Peter Stumpf, Managing Director, TransMit or Heinz Rupp. Prof. Maisch is the chairperson of "The Task Force on the Diagnosis and Management of Pericardial Diseases of the European Society of Cardiology": Guidelines on the Diagnosis and Management of Pericardial Diseases Executive Summary. Our mission
is to design, develop, and co-market new patented
technologies in the medical device field related to
the pericardial space. Each technology will fill a
current need in medical procedure by improving upon
an existing technology, or by designing a device to
serve a need that is clearly defined and
acknowledged by medical professionals. Major market segment. Cardiac Resynchronization Therapy Pacemakers (CRT-P). The resynchronization or biventricular pacing coordinates the beating of the left and right ventricles to work together efficiently and effectively pump blood throughout the body. The pacemaker can also pace the heart to treat slow or abnormal heart rhythms, i.e. standard pacemaker therapy. Patients with heart failure and intraventricular conduction delay may have a greater ability to complete physical activities and thus improve their quality of life. Advantages over currently available products. 1. truely minimal invasive access to the pericardial space. 2. no lung deflation that is a risk for most patients with an indication for CRT. 3. Intrapericardial mapping prior lead implantation for best improvement of pump function. Important for patients with prior myocardial infarction. 4. No competing device available that works for thickened pericardium. 5. Pioneering technology with prospects of future intrapericardial therapy interventions. Scope. CRT pacemakers are for use in patients who have moderate heart failure (NYHA Class III/IV). They are indicated for use in pateints who (i) exhibit symptoms related to heart failure (EF<= 35%) and QRS >= 120 ms) or (ii) remain symptomatic despite stable, optimal heart failure drug therapy. Contrary to conventional CRT, the left ventricular lead is not implanted via the coronary veins but is implanted in the epicardium via our Attacher/AttachGuider/AttachImplanter approach. The other leads are as in the conventional approach, i.e. one lead in the right atrium and one lead in the right ventricle. Also a regular titanium can containing a battery and electronic circuitry that provides multiple therapies is required. Clinical
advantages. The procedures
do not require lung deflation and permit
intrapericardial navigation.
Current state of the art technology. Epicardial leads are implanted currently in a surgical approach that requires the adverse lung deflation. Any procedure that does not require lung deflation is a major breakthrough. Procedures requiring lung deflation are no real alternative to transvenous CRT. Competitive comparison. A potentially competing product currently available relates to theStingray technique of Medtronic Inc. that to our knowledge requires lung deflation. The reasons for investing in a follow-up technology to coronary sinus CRT are given by Medtronic Inc: "Although bi-ventricular pacing has shown beneficial results, numerous problems are associated with this technique. One such problem is the amount of time required for a physician to insert the lead into the desired location on the left ventricle. Further, the placement of the lead on the left ventricle is limited to sites that provide adequate pacing and sensing signals. Further, cannulation of the coronary sinus is often difficult due to either the rotation of the heart or the presence of an enlarged right atrium or Thebesian valve. Further, the placement of a lead on the surface of the left ventricle though the coronary sinus cannot be carried out in some patients with prior bypass surgery or with coronary sinus stenosis. Finally, the coronary sinus lead provides an oftentimes unstable placement and can become detached after installation." It appears as a logical step to invest into a truely minimal invasive technology that avoids the risk associated with lung deflation. The Attacher/AttachLifter and AttachGuider represent thus an entirely new application with no current competition. Extensive experience has been accumulated with respect to the conventional access to the pericardial sac using a needle (1). This requires, however, pericardial effusion which clearly separates the epicardium from the pericardium and thus prevents puncture of the myocardium leading to life threatening cardiac tamponade. The needle technique is obviously not applicable to patients with normal pericardium but impaired heart function. This has clearly been stated by Prof. B. Maisch: "Whilst advocating the advantages of pericardiocentesis in appropriate cases of pericarditis, Dr Maisch points out that many cardiologists are fearful of using the technique unless severe cardiac tamponade is present, a situation in which, he estimates, the volume of the effusion is at least 500 ml. He said, “At Marburg we perform pericardiocentesis in the presence of as little as 100 ml of fluid, or sometimes even 50 ml. But I would not recommend this to the ‘normal’ cardiologist, as there is a high risk of damage to the heart. But I think the subxiphoidal approach should be used more widely, because it’s safer and makes it easier to perform a full aetiological diagnosis as well as to take off fluid.” Experience has been gathered during testing of the "Perducer" device of Comedicus Inc. which unfortunately can fail since it does e.g. not monitor the actual attachment of the pericardium before the puncturing step (8). The key technology is now available for accessing the pericardial space in a truely minimal invasive approach. The Attacher provides a solution to the critical step of tissue puncturing by monitoring tissue attachment before the puncturing step. The AttachLifter permits puncturing of thickened pericardium. The AttachGuider/AttachImplanter permit intrapericardial navigation with subsequent lead implantation. Growing evidence that resynchronization therapy can be and should be improved by lead placement which is not restricted by coronary veins or local conduction defects. In particular, apical pacing which is not possible with coronary sinus CRT appears to be superior. In a canine model of contraction dyssynchrony, left ventricular apical pacing appears to be a superior pacing site in the context of resynchronization therapy. This study supports the patents of Noel Gray (US 6,144,879, US 5,674,259) for pacing the heart with a plurality of electrodes positioned in the apical area of a heart. Research described
here is based on limited funding by the Marburg
Cardiac Society
and is not sponsored by medical device
companies. Please support
the projects by a donation through the
Marburg Cardiac
Society which is a non-profit
organization. For
details, please contact
Other web sites related to the repair of diseased heart maintained by us: www.cleverfood.com www.cardiorepair.com www.carditis.com www.herzzentrum-marburg.de herz.online.uni-marburg.de |
Our international patent applications:
Care has been taken to take into account all potential claims of the inventions as well as to protect them from possible competition from other technologies (including inferior ones). All patent application documents are available for examination by potential investors. Please note, the PCT applications still carry some "Xs". In the upcoming national applications, the claims will be narrowed down maintaining, however, the claims for our devices planned for production.
The patent applications can be downloaded as PDFs: Attacher.pdf, AttachLifter.pdf, AttachGuider.pdf

Copyright
H. Rupp is the owner of all content of this web site. The commercial use or publication of all or parts of the contents of this web site are forbidden in any form (e.g. books, magazines, CD-ROMs, WWW-pages) without the prior written permission of H. Rupp. The copyright for any material created by the author is reserved. Any duplication or use is not permitted without the author's agreement.
Impressum:
Dr. H. Rupp
Professor of Physiology
Experimental Cardiology Laboratory
Department of Internal Medicine and Cardiology
Heart Center
Philipps University of Marburg
Baldingerstrasse 1, 35043 Marburg, Germany
Tel. +49 6421 586 2775
Google Scholar Citations of H Rupp

November 23, 2013